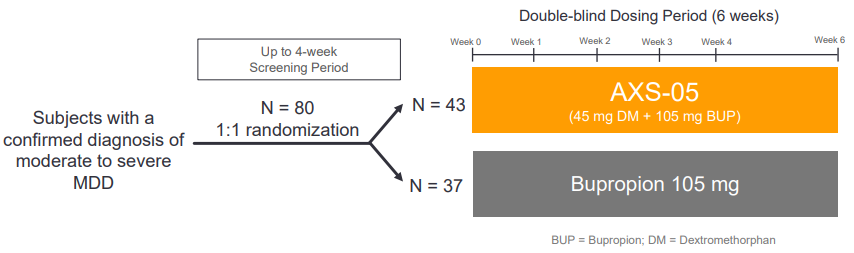
Breakthrough Therapy, PRIME and Sakigake: A Comparison Between Neuroscience and Oncology in Obtaining Preferred Regulatory Status - Elena Tomaselli Muensterman, Yijia Luo, Jonathon M. Parker, 2019

Jorge Luna on LinkedIn: So proud of my amazing team who won the Sage Therapeutics 2019 Field… | 59 comments
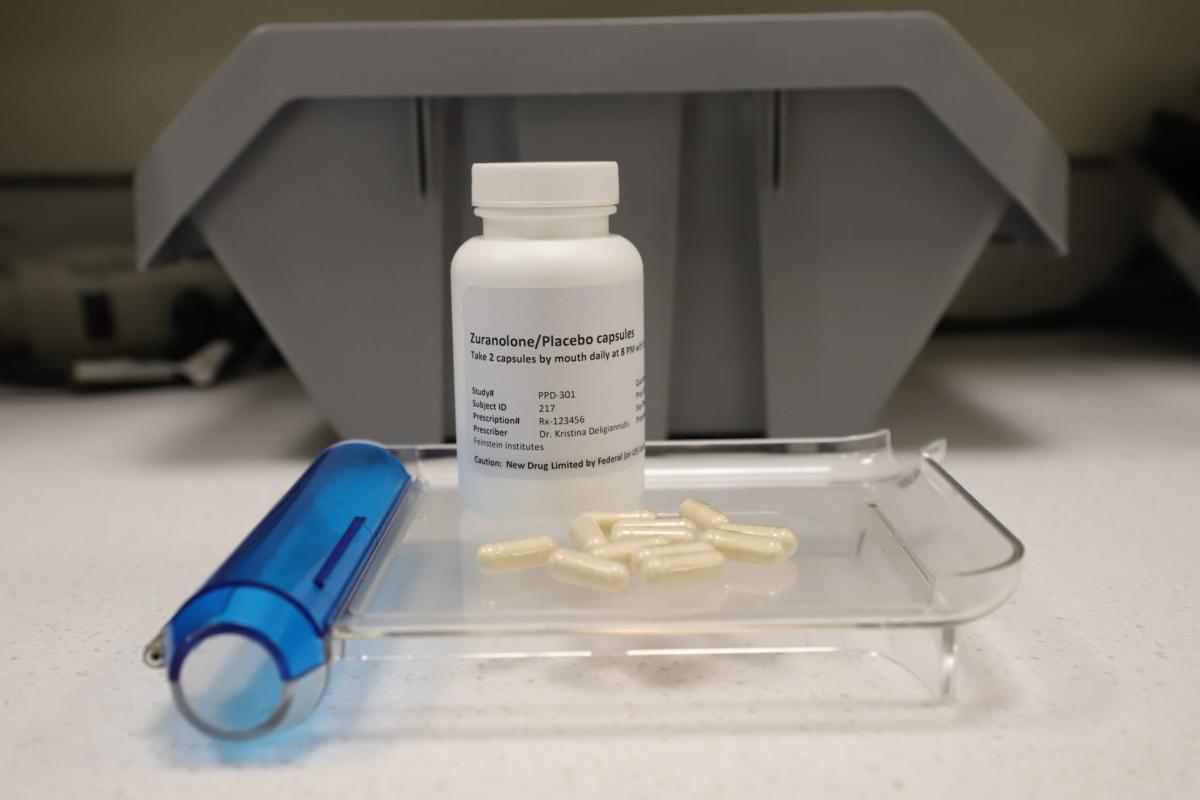
Zuranolone, a breakthrough postpartum depression treatment, was just approved by the FDA. The fast-acting pill can improve symptoms in 3 days
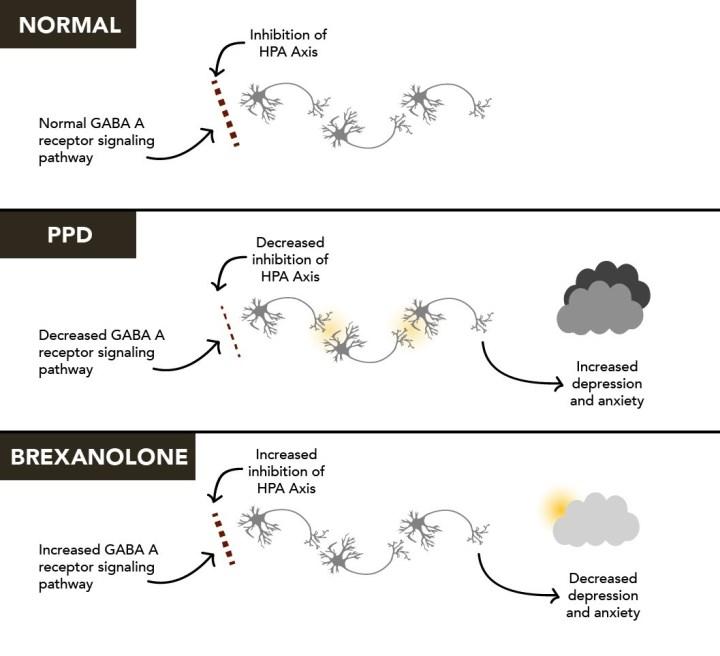
Sage Therapeutics: Zuranolone's Multi-Billion Dollar Market Opportunity More Than Justifies Company Valuation (NASDAQ:SAGE) | Seeking Alpha

Biogen and Sage Therapeutics Announce Global Collaboration to Develop and Commercialize Potential Breakthrough Therapies in Depression and Movement Disorders | Business Wire

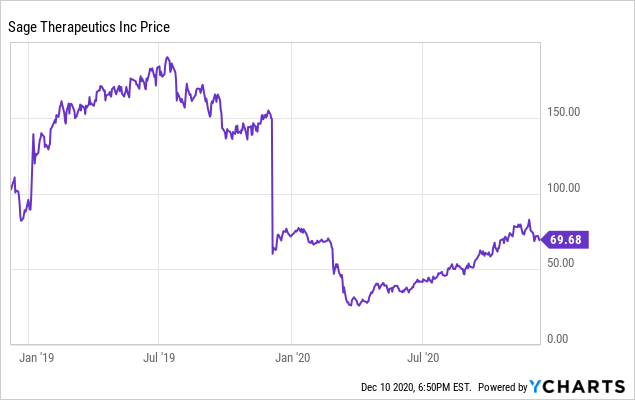
SY Investing on X: "$SAGE (-62% PM) announced pivotal Ph 3 MOUNTAIN results of SAGE-217 in MDD. Study didn't meet primary endpoint. #Fail SAGE-217 showed mean reduction of 12.6 in HAM-D total

Sage Therapeutics Announces Development Plan for Zuranolone (SAGE-217) Following Breakthrough Therapy Guidance Meeting with the U.S. Food & Drug Administration-CliniExpert