29Si NMR Spectroscopy as a Probe of s- and f-Block Metal(II)–Silanide Bond Covalency | Journal of the American Chemical Society

Suppose a pure Si crystal has 5 × 10^28 atoms m^-3 . it is doped by 1 ppm concentration of pentavalent As. Calculate the number of electrons and holes. Given that n1 = 1.5 × 10^16m^-3 .
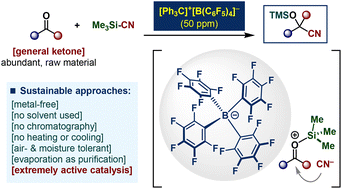
Sustainable organocatalytic cyanosilylation of ketones by PPM-level loading of triphenylcarbenium tetrakis(pentafluorophenyl)borate - Green Chemistry (RSC Publishing)

Solution State Structure Determination of Silicate Oligomers by 29Si NMR Spectroscopy and Molecular Modeling | Journal of the American Chemical Society